More than 85% of malignant tumors display high levels of telomerase activity. The unwinding of RNA quadruplex structures plays a key role in the regulation of telomerase function and provides therefore an attractive target for highly innovative anticancer therapies. In collaboration with Dr.
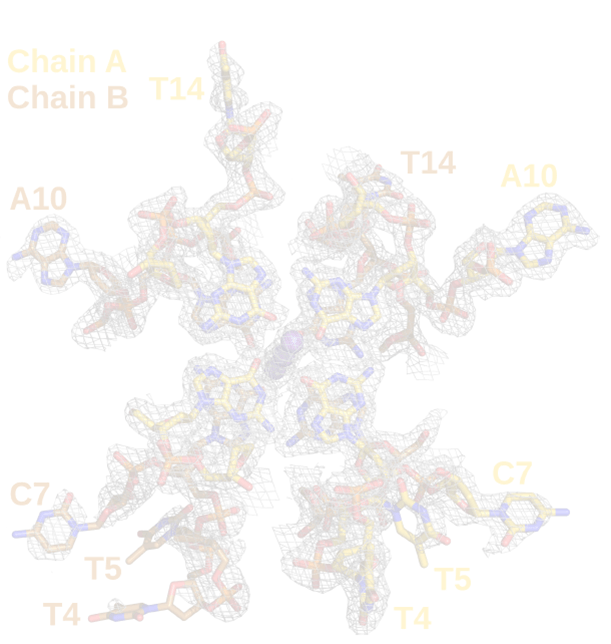
McKenna, we are deciphering the molecular mechanisms of RNA/DNA G-quadruplex unwinding by the human RNA-helicase machinery. The results demonstrate that precise networks of G-quadruplex recognition and unwinding are a major mechanism by which genetic mutations cause human diseases [1, 2]. The Stetefeld laboratory has unraveled the molecular mechanism that is required to bind G4-elements [3] by applying a combination of biophysical approaches (incl. CD, DLS and SAXS) in conjunction with NMR techniques. This work describes the first high-resolution study of G4 recognition, and is expected to have significant impact on the design of small-molecule stabilizer/inhibitors of RNA/DNA-quadruplex helicases. Recently, we reported the first high-resolution X-ray structure of a unique unimolecular DNA G4 with a cytosine bulge that alters the surface of the DNA, providing an opportunity to develop drugs that can specifically target individual G4’s [4].
[1] Booy et al. Nucleic Acid Research, 2012 [2] Booy et al. Nucleic Acid Research, 2014 [3] Meier et al. JBC, 2013 [4] Meier et al. Nucleic Acid Research, 2018
